Covered by insurance for the patient as a pharmacy benefit in the first 12 months
Conception Kit® Description
A three-month treatment prescription-only medical device for at-home use. The kit is designed to enhance success rates of conception and is FDA 510K cleared.
Approved Indications:
1) Low sperm count
2) Low sperm motility
3) HVE (Hostile Vaginal Environment [i.e., acidic pH])
The kit contains the following items for 3 monthly cycles:
3 FDA-cleared Conception Caps®
1 Practice Conception Cap®
24 Conceivex Ovulation Predictors
3 Latex-Free Conceivex Semen Collectors
3 Sperm-Friendly Intimate Moisturizers
3 Pregnancy Tests
1 Conception Wheel
1 Conception Journal
1 Instruction Manual
The Conception Cap®
- FDA-cleared cervical cap for at-home use
- Concentrates all the available semen, unexposed to the vaginal environment, at the opening of the cervical os
- Made of soft, flexible, implantable-grade silicone
- Designed with a built-in loop for easy removal
- The soft dome collapses after placement, putting sperm cells in direct contact with the cervical mucus for 4-6 hours. Patient is free to move around once cap is in place.
Semen Collector
- FDA-cleared semen collection device designed to aid conception
- Similar to a condom and used to capture and transfer semen to the conception cap after coital relations
- Lab tested for cervical mucus and sperm compatibility
- Proven to not modify the properties of semen and cervical mucus
Using the Kit – All Steps Performed by Patient at Home1. Planning and practice
2. Cervical-cap insemination
3. Test Pregnancy: Use included pregnancy tests and schedule a doctor visit if conception is achieved |
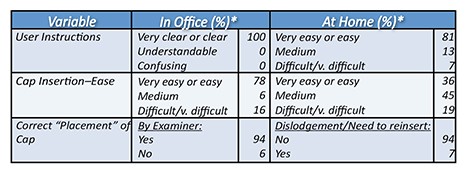
Registration Clinical Trial Objective: Determine patient’s ability to understand the written instructions, manipulate the condom and fill the cap, place the cervical cap on the cervix without spilling the content, and remove when ready, determine that the materials did not change the cervical mucus or sperm in any way.
Patients: Participating women (ages 18-42) used the product in a physician office (n=32) or at their homes (n=31) after receiving instructions from a physician. Method: Success was defined as the woman being able to demonstrate her knowledge of the process by correctly responding to a questionnaire. Primary endpoint:
|
Studies done on cervical cap. For FDA clearance, Conceivex is claiming the class.
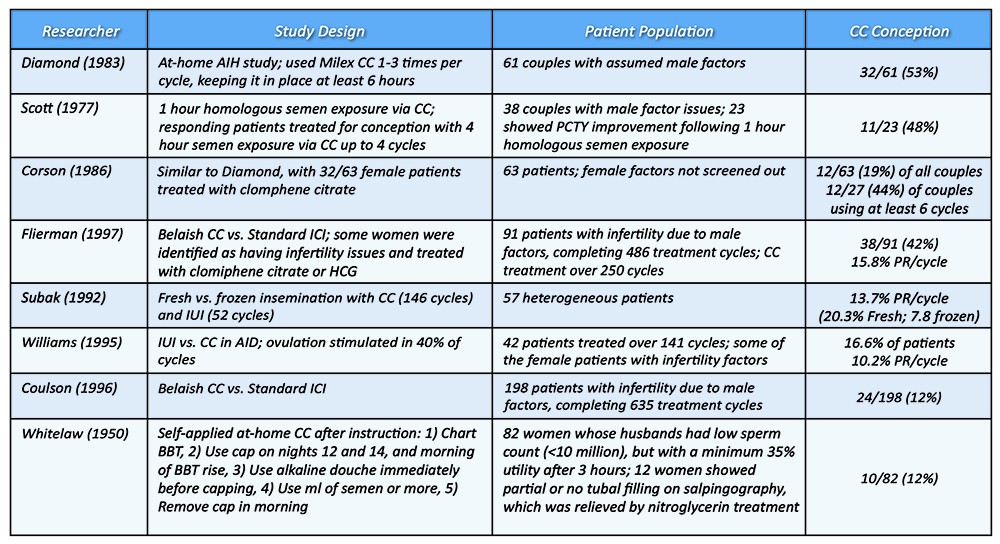
Source: Rivet EL. Cervical caps for conception: clinical data, empirical evidence & limits in research. Linacre Quarterly. 2010. 77 (1): 30-42
CC = Cervical Cap